And yes, we ignore the noble gases for EN values because they are happy as is - they have no desire for any shared electrons and they don't form bonds, so no values for them. We use it the most of the three trends/properties last listed. Think of EN as the "pull" on electron pairs in a molecule by an atom. The minimum of 0.8 on this scale belongs to cesium (bottom left). The maximum of 4.0 on this scale belongs to fluorine (top right). Electronegativity is the purposeful human friendly scale from 0 to 4 that electron affinity lacked. Move on to electronegativity now.Įlectronegativity is a relative scale from zero to four that measures the "desire" or "pull" on electron pairs. I'm including this for the purpose of pointing out this is a real measurement and the recognition of EA is more important for our studies than the actual values. The trends on the periodic table are not as pronounced as with other trends (they're a bit janky) - but in general, the upper right corner has the largest EAs while the lower left corner has the lowest values. Less desire is smaller energy and there is even no desire and the numbers go to zero and even negative. If the atom "wants" the electron a lot, then the EA is big. You can think of EA as the "desire of an electron" by an atom. 
Going across rows, IE's increase.Įlectron affinity is the amount of energy released when one electron is added to a neutral atom (A) in order to form a –1 anion. Much like all the trends, the two extremes of this property are at the bottom left (smallest IE) and the top right (largest IE). All atoms have a wide variety of energies needed to do this, but they DO follow a trend that is easily seen on the periodic table. The energy needed to do this must overcome the attraction of the outermost electron to the nucleus. Ionization energy is the amount of energy it takes to remove one electron from a neutral atom (A) in order to form a +1 cation. This is illustrated below starting on the left with a neutral atom. And, each subsequent removal of additional electrons leads to smaller and smaller cation species. This means that cations have smaller radii than the neutral atom from which they came from. Upon each e – removal, there are fewer e – repulsions which means the remaining electrons are pulled in tighter than before. Realize that when you make a cation from a monatomic neutral species, you are removing electrons from the outmost valence shell. The typical number is one to three electrons to make +1, +2, and +3 cations. Now that you have the trend for neutral atoms, let's modify or tweak those sizes for when the atom is changed into a cation or anion.Ĭations: Metals tend to lose their electrons to make stable cations. Below is a simple graphic illustrating the atomic radii trends. So on any one row, the group 1 atoms (alkali metals) are the biggest on that row and the group 18 atoms (noble gases) are the smallest. However, as you go across, the nuclei are getting more and more positive (more protons) - therefore there is more + to – attraction and the electron cloud is pulled in tighter and therefore a smaller radius. The logic is that as you go across rows, you are staying in the same main energy level (n) so electrons are entering the atomic atmosphere at about the same distance. This may seem counterintuitive but it is the fact. This is because in going down a column you are jumping up to the next higher main energy level (n) and each energy level is further out from the nucleus - that is, a bigger atomic radius.Ītoms get smaller as you go across a row from left to right. 
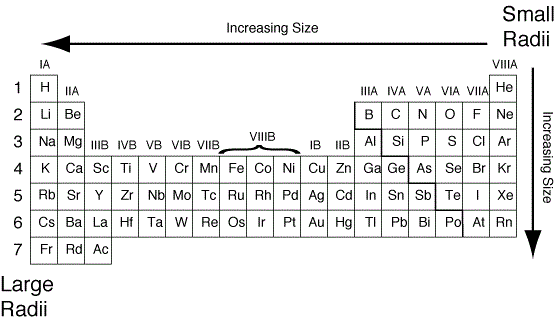
so chopping that to 50-300 pm isn't a big deal.Ītoms get bigger as you go down a column on the periodic table. Oh well, you should just wrap your head around the general range of all atomic sizes the extremes are 31 pm and 343 pm. Sometimes we just do a generalized bit of rounding as well and say things like atoms range from about 50 pm to 300 pm which is more of a 6:1 ratio. So that is roughly a 10:1 ratio of largest to smallest. Which atom is the largest? That would be cesium, Cs, which comes in with a radius of 343 pm. Yeah, He is even smaller than hydrogen, H, which is 53 pm. The smallest atom on the periodic table is helium, He, and has a radius of 31 pm. All our trends describe the trend in two directions on the periodic table: 1) across a row, and 2) up and down a column. A trend is generally "it gets bigger" or "it gets smaller" sort of thing. We use the periodic table to help us recognize certain trends of physical and chemical properties of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
